
How Do Industrial Centrifuges Work?


You know that a centrifuge either removes solids from liquids or separates two liquid phases from each other by spinning them at high speed—and thus harnessing centrifugal force.
But how—and why—does centrifugation cause materials to separate?
How Do Centrifuges Work? By Harnessing Stokes’ Law
If you want to meaningfully understand your centrifuge, it’s best to start with a quick review of Stokes’ Law. This was first expressed more than 150 years ago by George Gabriel Stokes. And, while it might not be immediately apparent from the textbook description (“Use this formula to calculate the frictional force—aka drag force—a viscous fluid exerts on a spherical object with a very small Reynolds”), it’s still instrumental in determining how quickly materials will separate.
According to Stokes, there are just five factors that contribute to how quickly particles will settle out of a fluid:
- particle size
- particle density
- fluid density
- fluid viscosity
- the force of gravity
All else being equal, Stokes correctly predicts the following settling times in water:
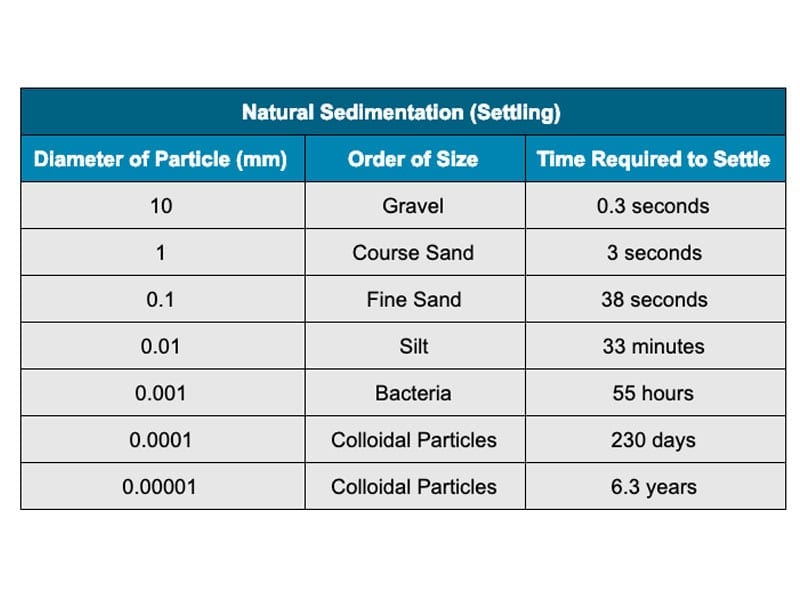
Six years is a long time to wait for clean coolant. But if we can control just one of those five factors—particle size or density, fluid viscosity or density, or the force of gravity—then we can take control of the entire settling process. Instead of waiting six years for particles to settle out on their own, we can pull them out in just minutes.
Centrifugation vs. “Artificial Gravity”
When spinning at high RPMs, it’s not at all unusual for the fluids in a centrifuge’s rotor to experience more than 13,000 times the force of gravity. In essence, we’re creating artificial gravity in the rotor–from the fluid’s perspective. That gives us control of one factor in Stokes’ equation and massively speeds the settling process.
But this isn’t just about speeding up the settling process. A spinning centrifuge forces denser materials to its outer edge (causing the less dense materials to flow toward the center of the rotor). By carefully tuning the shape of the rotor, interior structures of the rotor, and the speed at which it spins, we can separate fluids and solids with a surprising degree of accuracy and efficiency—even separating multiple liquids and solid phases in a single pass.

Client-Site Services
Committed to Service
With the largest fleet of separation technology, advanced engineering, and chemistry capability across a wide array of industries and experienced operations personnel throughout North America, you can count on us when you need us most.
- 9 Service Locations Across North America
- The Largest Fleet of Separation Technology
- Over 150 of Fluid Experience





